Faces form during the very early stages of embryology. from www.shutterstock.com
Is your face long? Wide? Big nose? Small ears? High forehead?
It’s our faces that characterise how the world sees us, and how we recognise our close friends and family. If you’re lucky enough to be born with a highly symmetrical or a very unique face, perhaps you might have a career as a model or actor.
But how do our faces come about – and what happens when things go awry? We need to look way back to the early stages of life to find out.
From a fertilised cell
Like humans, most creatures throughout the animal kingdom have an instantly recognisable face. Such distinctive features as the trunk of an elephant, the long jaws and abundant sharp teeth of a crocodile, varied shapes and sizes of bird beaks and the unique bill of the platypus are all distinct and recognisable.
Our faces arise during the earliest stages of life. And quite incredibly, the processes that give rise to all these distinctive faces – animal and human – are exceptionally well conserved (that is, haven’t changed much over the course of evolutionary history). Amongst humans and other creatures with backbones (together known as vertebrates), the genes and biological processes that make a face are really very similar.
All animals and humans start out as a fertilised cell. Through thousands of cell divisions, the tissues that will eventually make up the skull, jaws, skin, nerve cells, muscles and blood vessels form and come together to create our face. These are the craniofacial tissues.
The face is among the earliest recognisable features that form in an embryo, with the future eye, nose, ear and tissues that will eventually form the upper and lower jaws all established by about 7-8 weeks in human gestation.
Fusion of two sides
By the sixth week of human development, the major fusion processes of the face have taken place – the two sides of the developing nose will join, both to each other and to the tissue that will become the upper lip. This first fusion (the formation of the “primary palate”) establishes the correct anatomy of the face, and serves as a structural guide for the next major fusion event – that of the secondary, or hard palate.
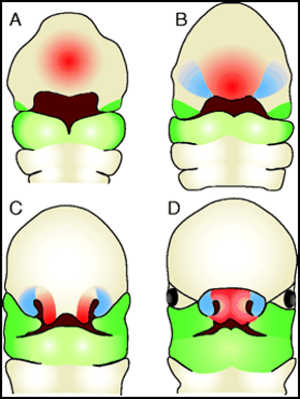 The formation of the face – tissues that comprise the future nose and upper lip (red), the sides of the nose (blue) and the upper and lower jaws (green) arise by the 4th week of development (A) and have migrated and fused to form a distinctive ‘face’ by the 8th week of development (D). New insights into craniofacial morphogenesis, CC BY
The formation of the face – tissues that comprise the future nose and upper lip (red), the sides of the nose (blue) and the upper and lower jaws (green) arise by the 4th week of development (A) and have migrated and fused to form a distinctive ‘face’ by the 8th week of development (D). New insights into craniofacial morphogenesis, CC BY
The hard palate originates as two separate “shelves”, one from the left side of the embryo and one from the right. These shelves elevate and grow together to form one continuous structure, ultimately separating the cavities of the nose and sinuses from that of the mouth. (You can feel this hard palate with your tongue – it’s the roof of your mouth.)
Once these fusion processes are complete (by about week 9 of gestation, still well inside the first trimester), the cells of the face still continue to dynamically move, reshape, and take on functional roles. This includes forming the structural framework of the bones, the delivery of oxygen and nutrients by the blood vessels, and controlling eye and jaw movements by the facial muscles.
Sometimes things go astray
Of course, given the incredible complexity and synchronicity required for all these cells and tissues to end up in the correct space, it is perhaps very surprising that things do not go wrong in craniofacial development more often than they do.
Across the world, 4-8% of all babies are born each year with defects affecting one or more organs. Of these children, 75% show some anomaly of the head or face.
Problems can occur with any cell types that make up the skull, face, blood vessels, muscles, jaws and teeth.
But one of the most common craniofacial defects are palatal clefts, where the hard palate does not fuse correctly, leaving children (roughly 1 in 700 worldwide) with a large gap between their nasal passages and mouth.
Although relatively easily corrected by trained reconstructive surgeons in first-world health care systems, significant ongoing healthcare is still essential.
Services such as speech pathology and psychological counselling are often required. The children also may need medical attention to improve hearing, as problems with middle ear bones often come with other craniofacial defects.
Later surgeries to correct muscular defects do not come cheaply – assuming of course that such surgical and allied health is available to the individual in the first place. This is frequently not the case outside the first world.
Understanding why problems occur
To reduce both the severity and incidence of craniofacial defects, researchers use animal model systems – particularly mouse, chicken, frog and zebrafish embryos – to try and uncover the reasons why these defects occur.
Of all craniofacial defects, 25% are attributed (at least partially) to environmental factors such as smoking, heavy alcohol or drug use, toxic metals and maternal infection (such as salmonella or rubella) during pregnancy.
About 75% of all craniofacial defects are linked to genetic factors. As most of the genes that control craniofacial development in animals also do so in humans, using these animal models helps us better understand human palate development and how specific genes are involved.
Eventually this work may lead to new prevention and treatment strategies, for example supplementing the mother’s diet with beneficial nutrients and vitamins.
An example of such an intervention is the B-vitamin folate, used to reduce neural tube defects such as spina bifida. Mandatory folic acid fortification of food in the USA in 1999-2000 resulted in a 25-30% reduction in severe neural tube defects, clearly an exceptional outcome for newborns and their families.
Through greater understanding of the genetic processes that drive facial growth, further beneficial factors will be identified that can be safely given to pregnant mothers, and give a far better start to life to children that may otherwise be born with a craniofacial disorder.![]()
About The Author
Sebastian Dworkin, Group Leader, Developmental Genetics Lab, La Trobe University
This article is republished from The Conversation under a Creative Commons license. Read the original article.
Related Books
at InnerSelf Market and Amazon