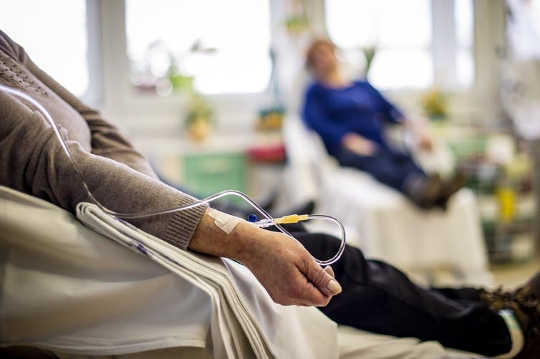
Many cancer patients have chemotherapy after surgery, but not all of them actually need it. shutterstock.com
Many cancer patients could soon be spared the unnecessary side effects of chemotherapy after having surgery to remove their tumour. A blood test being trialled at more than 40 hospitals across Australia and New Zealand aims to detect whether there are any cancer cells remaining in the body after surgery, which could lead to the cancer returning.
There is currently no reliable way of knowing which patients will have their cancer return after surgery. So, early stage cancer patients often receive chemotherapy after surgical treatment as a precaution – to mop up any cancer cells that could remain.
But chemotherapy comes with a host of serious side effects. Short-term, these include pain, fatigue, nausea and other digestive issues, bleeding problems and an increased susceptibility to infection. Long term side-effects may include heart, lung, nerve and memory problems, and fertility issues.
When cancer cells rupture and die – which they are always doing – they release their contents, including cancer-specific DNA, that float freely in the bloodstream. This is called “circulating tumour DNA” or ctDNA. If ctDNA is detected after surgery, this indicates there are remaining microscopic cancer cells in the patient that weren’t picked up by standard tests.
Research shows patients positive for circulating tumour DNA after surgery have an extremely high risk of cancer relapse (close to 100%) while those with a negative test have a very low risk of relapse (less than 10%).
Current trials in early-stage bowel cancer patients started in 2015. These have shown the ctDNA test can determine whether patients can be divided into “high risk” and “low risk” groups. The trials were later extended to women with ovarian cancer in 2017 and will soon extend to pancreatic cancer.
Results from the same test could also help scale the dose for the patients who do need chemotherapy, depending on their risk of cancer returning.
Why do we need the test?
When a patient with a cancer, such as early stage bowel cancer, is diagnosed, their tumours seem to be limited to the bowel with no evidence of spread to elsewhere in the body. But after a successful surgery to remove the bowel cancer, around one-third of these patients will experience cancer recurrence elsewhere in the body in the following years.
This shows that cancer cells have already spread at the time of diagnosis, but could not be detected using our current standard blood tests and scans. If these patients had been treated with chemotherapy after surgery, these relapses would have been prevented by eradicating the microscopic residual cancer cells responsible for the cancer’s return.
In the case of bowel cancer, the decision on whether to use chemotherapy is based on an assessment of the cancer removed at the time of surgery in the lab. For example, if there are cancer cells in the lymph glands next to the bowel (a stage 3 cancer), there is an increased probability the cancer has already spread elsewhere.
For other cancers, such as ovarian and pancreatic, other methods are used to determine whether chemotherapy is necessary. But they all lack precision. Ultimately, some high-risk patients will not have cancer recurrence because their cancer has been cured by surgery alone, while other apparently low-risk patients will suffer from recurrence.
So, many bowel cancer patients are currently treated with six months of chemotherapy and its associated side effects, even though they do not need to be treated. While others that would potentially benefit from treatment do not receive the necessary chemotherapy because they appear to be at low risk.
More than 400 patients have already joined in the trials but there is hope this will grow to more than 2,000. The trials are expected to run until 2021 for bowel cancer and 2019 for ovarian cancer.
The ctDNA test was developed through a collaboration between the Walter and Eliza Hall Institute and Johns Hopkins Kimmel Cancer Centre, US.
The ability to find and measure cancer DNA in patient’s blood could revolutionise cancer care. The next step is to determine how it can be used in the clinic.![]()
{youtube}https://youtu.be/xMHPAh7g_qI{/youtube}
About The Author
Jeanne Tie, Associate Professor, Walter and Eliza Hall Institute
This article is republished from The Conversation under a Creative Commons license. Read the original article.
Related Books
at InnerSelf Market and Amazon